An Authorization Is Required for Which of the Following Hipaa
Sale of protected health information. According to HIPAA requirements outlined in.

The Hipaa Minimum Necessary Standard Compliancy Group
1 To the Individual unless required for access or accounting of disclosures.

. A HIPAA authorization has which of the following characteristics. The Authorization must be written in plain language. When can protected health information be disclosed without authorization.
To others who are required by law to review the quality and safety of the research including. Authorization should be obtained in writing from prospective subjects. I Notwithstanding any provision of this subpart other than the transition provisions in 164532 a covered entity must obtain an authorization for any disclosure of protected health information which is a sale of protected health information as defined in 164501 of this subpart.
Uses and Disclosures for research Purposes and 2 HIPAA-32 policy. This will allow the researcher to use. Authorizations should have certain elements to be considered valid.
Use or disclosure of PHI for marketing purposes except when communication occurs face to face between the covered entity and the individual or when the communication involves a promotional gift of nominal value. 1 Open form in Adobe Reader. Does the HIPAA Privacy Rule require documentation of Institutional Review Board IRB or Privacy Board approval of an alteration or waiver of individual authorization before a covered entity may use or disclose protected health information for any of the following provisions.
An authorization in HIPAA terms is the consent of an individual or patient providing explicit authorization to use or disclose their personal information. What is the general rule for patient authorization. Question 9 - Which of the following is NOT true regarding a Business Associate contract.
The authorization must also include a statement about any direct or indirect remuneration the covered entity has received or will receive from a third party. If granted by the IRB the Alteration of Authorization allows the researcher to omit one of the core elements of a valid authorization. One of the core elements of a valid authorization under HIPAA is the signature of the individual 45 CFR 164508cvi.
Section 164508 c 1 i. Under HIPAA retrospective research aka data mining on collections of PHI generally. 3 An authorization to use or disclose psychotherapy notes may not be combined with an authorization to disclose other forms of PHI.
In addition the authorization must state that the covered entity is. An authorization for marketing purposes can be included on the organizations compliant HIPAA authorization form or a separate one may be created. A HIPAA authorization is consent obtained from an individual that permits a covered entity or business associate to use or disclose that individuals protected health information to someone else for a purpose that would otherwise not be permitted by the HIPAA Privacy Rule.
2 Treatment Payment and Health. Uses and Disclosures Requiring Patient Authorization. The HIPAA Privacy Rule requires that an individual provide signed authorization to a covered entity before the entity may use or disclose certain protected health information PHI.
A valid authorization must contain certain required statements. Under the Privacy Rule and in accordance with the minimum standards doctors nurses hospitals laboratory technicians and other health care providers that are covered entities may. Uses plain language that the data subject can understand similar to the requirement for an informed consent document.
To be valid a HIPAA authorization must satisfy the following 2. A specificmeaningful description of PHI that will be collected for research and the purpose of collecting this information eg for this research 2. The authorization may not be combined with any other document such as a consent for treatment.
Use or disclosure of PHI otherwise not permitted by the HIPAA Privacy Rule. HIPAA authorization is required for. All of the following are true about Business Associate Contracts EXCEPT.
The name or other specific. A covered entity must get patient authorization to sell an individuals protected health information PHI. Receives a valid Authorization for its use or disclosure of PHI for research it may use or disclose the PHI for the research but the use or disclosur e must be consistent with the Authorization.
Under HIPAA the following core elements and statements must be. Valid HIPAA Authorization Requirements. Any suspected or confirmed breach or loss of PHI will be immediately reported to the IRB and HIPAA Privacy Office.
1 for preparatory research at 45 CFR 164512 i 1 ii 2for research on the protected health. Is required between a Covered Entity and Business Associate if PHI will be shared between the two Is a written assurance that a Business Associate will appropriately safeguard PHI they use or have disclosed to them from a covered entity. Whether combined with an informed consent or separate an Authorization must contain the following specific core elements and required statements stipulated in the Rule.
A HIPAA authorization can allow a covered entity to use or disclose an individuals PHI for its own research purposes or disclose PHI to another entity for that entitys research activities. Thus revocation of an authorization limits a covered entitys own continued use of the health. A description that identifies the requested information in a specific and meaningful fashion 45 CFR.
Both Covered Entities and Business Associates are required to ensure that a Business Associate Contract is in place in order to be compliant with the HIPAA regulations. A covered entity is permitted but not required to use and disclose protected health information without an individuals authorization for the following purposes or situations. Could not practicably be conducted if the research participants authorization were required or to access existing databanks or repositories.
The IRB may alter or waive researchparticipants authorization for use or disclosure of PHI for research provided that. In order to be accepted by doctors and hospitals a HIPAA release authorization must have six core requirements. 2 Use Fill Sign tool to complete.
The HIPAA rules and regulations consists of three major components the HIPAA Privacy rules Security rules and Breach Notification rules. An Authorization can be combined with an informed consent document or other permission to participate in research. Business Associates are required to ensure that Business Associate.
UC HIPAA Research Authorization 2014 Page 1 of 4 UC Health Version 2016 HIPAA Research Authorization. Is research and so requires either an authorization or meeting one of the criteria for a waiver of. 45 CFR 164508 researchers should obtain written authorization from subjects before using or collecting protected health information PHI whenever possible.
In this case the signature of the participant. If the ICF includes HIPAA authorization or if stand-alone HIPAA authorization is used the following elements are required. Read on to see what those items include.
You are allowing UCSF Health to release the following.

Hipaa Frequently Asked Questions Faqs The University Of Oklahoma Health Sciences Center

Hipaa Authorization Requirements Consent To Disclose Phi

Hipaa Authorization Requirements Consent To Disclose Phi

Hipaa Authorization Requirements Consent To Disclose Phi

The Following Slides Explain The Required Elements That Must Be Included For A Hipaa Authorization To Be Valid Hipaa Authorizations Ppt Download

Employee Sample Survey Form Survey Form Sample Survey Survey Template
Hipaa Deep Dive Series When Is A Patient Authorization Not Needed For Sharing Personal Data Polymer

Microneedling Consent Form Spa Salon Esthetician Etsy Consent Forms Microneedling Salons

Researching The Appropriateness Of Care In The Complementary And Integrative Health Professions Part 2 What Every Researcher And Practitioner Should Know About The Health Insurance Portability And Accountability Act And Practice Based Research

What Is Hipaa Compliance Hipaa It Requirements Dnsstuff
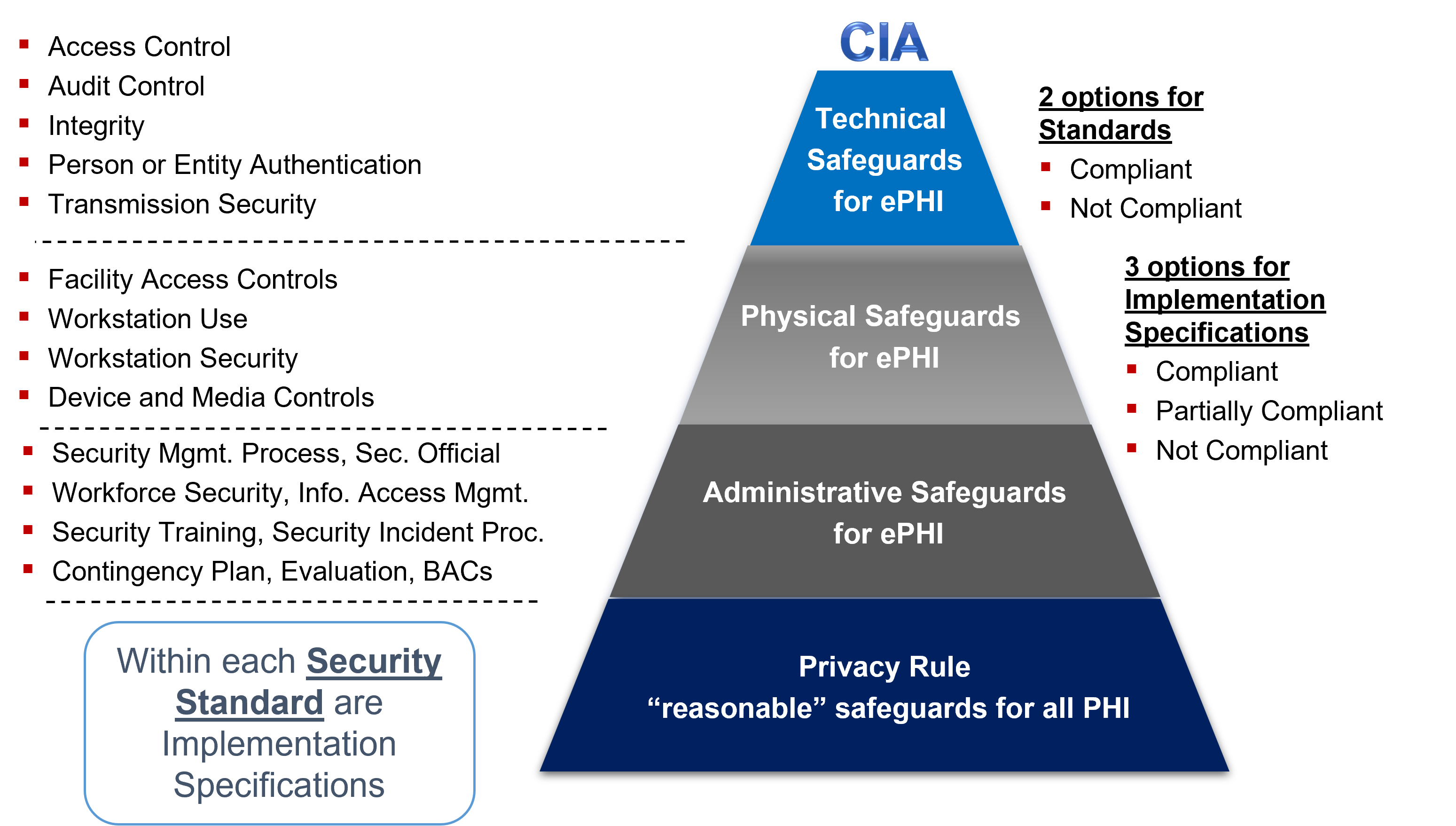
Hipaa Security Rule Hipaa Academy Beyond Hipaa Hitech Mu Ehr Health It Health Insurance Portability And Accountability Act

10 Times Hipaa May Not Apply Emergency Physicians Monthly

Hipaa Authorization Requirements Consent To Disclose Phi

Employee Sample Survey Form Survey Form Sample Survey Survey Template




Comments
Post a Comment